Abstract
Background Approximately twenty percent of patients with newly diagnosed acute myeloid leukemia (AML) have TP53 mutations, associated with complex karyotype and the highest risk of relapse(Döhner et al., 2017; Giacomelli et al., 2018). Although combination therapies, such as Ara-C + anthracyclines and Venetoclax (VEN) + hypomethylating agents (HMA) result in higher remission rates in patients with TP53-mutant AML, the mutant clone is never eliminated and invariably leads to relapse(Short et al., 2021; Takahashi et al., 2016). Novel targets and mechanisms associated with TP53-mut AML need to be identified.
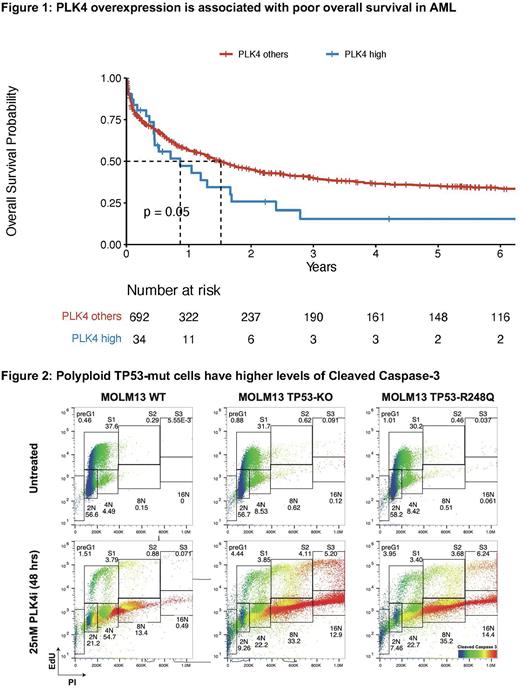
Results: We performed RNA-sequencing (RNA-seq) on FACS-sorted subpopulations of TP53-mutant or TP53-wild-type (wt) high-risk AML patients before and after treatment. Amongst overexpressed genes, we identified Polo-like kinase 4 (PLK4), a key regulator of centriole biogenesis and therefore of polyploidy, as a potential target highly expressed in TP53-mutant AML using differential gene expression analysis of TP53-wt vs TP53-mutant samples collected (TP53-mutant=36, TP53-wt=31, PLK4 LOG2FC=~2, p<0.001). We validated this finding using an independent RNA-seq dataset from the Munich Leukemia Lab of 726 AML samples (TP53-mutant=72, TP53-wt=654, PLK4 LOG2FC=0.48, p<0.001) and investigated the impact of PLK4 expression levels on AML patient outcomes in the context of TP53-status. High levels of PLK4 expression in AML are associated with poor outcomes (Figure 1).
Targeting PLK4 by CFI-400945 (25nM, Treadwell Therapeutics, Toronto, Canada) in MOLM-13 cells with CRISPR engineered knockout/mutations of TP53 for 72 hours resulted in increased polyploid cells in TP53-mut cells compared to TP53-wt AML (> 2 fold, p< 0.0001). PLK4 inhibition triggered higher levels of cleaved caspase 3 in polyploid TP53-mut polyploid MOLM-13 cells compared to TP53-wt MOLM-13 cells (Figure 2). Polyploidization in TP53-mut MOLM-13 cells 48 hours post treatment with 15nM of PLK4 inhibitor was not reversible and retained higher apoptogenic activity in TP53-mut MOLM-13 cells even after wash out for additional 48 hours.
To translate our findings to in vivo models, we transplanted CRISPR-generated TP53-wt, TP53-ko, and TP53-248 mutant MOLM-13 cells (Boettcher et al., 2019) into NSG mice (n=58: untreated n= 28, treated n=30), confirmed successful engraftment, and treated the mice with CFI-400945 (oral gavage 7.5 mg/kg, 5 times/week) for 21 days. Our preliminary data indicates that all treated mice showed improved survival compared to untreated mice. The median survival for treated TP53-wt vs untreated TP53-wt was 17.5 vs 14.5 days (20.7% increase, n=20 (untreated n=10, treated n=10), p=0.00086). The median for treated TP53-R248Q vs untreated TP53-R248Q was 15.0 vs 12.0 days respectively (25% increase, n=19 (untreated n=9, treated n=10), p=0.00039). The median OS for treated TP53-ko vs untreated TP53-ko was 11 vs 8 days respectively (37.5% increase, n=19 (untreated n=9, treated n=10), p<0.0001).
Next, we performed a Cox proportional hazard model to investigate the contribution of TP53 genotype and PLK4 inhibition on overall survival. The resulting model (Likelihood ratio test=83.09 on 3 degrees of freedom and p<2.2e-16) showed a higher hazard ratio in recipients with TP53-R248Q and TP53-ko (~4.3, and ~1.2 hazard coefficients respectively) in comparison to TP53-wt (p=2.67e-14, and 0.000708 respectively). Additionally, treated mice had statistically significant lower hazard ratios compared to untreated (hazard coefficients=-2.47277 and p=8.38e-10). The data suggest that PLK4 inhibition is a promising treatment approach for AML with TP53 mutation. We are presently testing different dosing schedules in other aggressive TP53-mut AML models to further ascertain the efficacy of PLK4 inhibition.
Conclusion: We established an association between PLK4 overexpression and poor survival outcomes in patients with newly diagnosed AML. TP53-mutant AML express higher levels of PLK4 in comparison to TP53-wt AML and targeting PLK4 induces polyploidy and apoptotic cell death predominantly in TP53-mutant AML. We demonstrate that targeting of PLK4 improves overall survival in TP53-mut and TP53-ko AML cells in vivo. Data suggest that targeting PLK4 represents a potential treatment approach for TP53-mut AML. An early phase, multi-center clinical trial is ongoing with CFI-400945 in AML (NCT04730258, TWT-202).
Disclosures
Nishida:Daiichi-Sankyo Inc.: Research Funding; Kintor Pharmaceutical: Research Funding. Walter:MLL Munich Leukemia Laboratory: Current Employment. Bray:Treadwell Therapeutics: Current Employment. Issa:Celgene, Kura Oncology, Syndax, Merck, Cullinan and Novartis: Research Funding; Novartis, Kura Oncology, Nuprobe: Consultancy. Haferlach:Munich Leukemia Laboratory: Current Employment, Other: Part ownership. Borthakur:Astex Pharmaceuticals, Ryvu, PTC Therapeutics: Research Funding; Pacylex, Novartis, Cytomx, Bio Ascend: Membership on an entity's Board of Directors or advisory committees; Catamaran Bio, Abbvie, PPD Development, Protagonist Therapeutics, Janssen: Consultancy. Andreeff:Chimerix: Current holder of stock options in a privately-held company; Oncolyze: Current holder of stock options in a privately-held company; Reata: Current holder of stock options in a privately-held company; CLL Foundation: Membership on an entity's Board of Directors or advisory committees; NCI: Membership on an entity's Board of Directors or advisory committees; German Research Council: Membership on an entity's Board of Directors or advisory committees; Leukemia & Lymphoma Society: Membership on an entity's Board of Directors or advisory committees; Cancer UK: Membership on an entity's Board of Directors or advisory committees; Medicxi: Consultancy; Glycomimetics: Consultancy; Aptose: Consultancy, Membership on an entity's Board of Directors or advisory committees; Senti Bio: Consultancy, Research Funding; Syndax: Consultancy, Research Funding; Brooklyn ITX: Research Funding; Pinot Bio: Research Funding; Oxford Biomedical UK: Research Funding; AstraZeneca: Research Funding; Breast Cancer Research Foundation: Research Funding; Daiichi-Sankyo Inc.: Consultancy, Research Funding; Kintor Pharmaceutical: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal